The Headline Trial
Currently Recruiting
26 Mar 2026
Up to $27,460
Melbourne, Australia
We’re currently looking for adults aged 18 to 65 who are experiencing androgenetic alopecia (AGA), also known as pattern hair loss, to take part in The Headline Trial.
Hair loss is common and can affect both men and women. This clinical study is investigating new approaches that may help shape future treatment options.
You don’t need a formal diagnosis to get started. If you think you may have AGA, you can begin with a quick pre-screen. This helps us assess whether you may meet the initial study requirements. Completing the pre-screen doesn’t guarantee enrolment, but it’s an important first step in seeing whether the study may be suitable for you.
By getting involved, you’ll be contributing to research that aims to improve future options for people affected by hair loss.
Join our clinical trial
We’re currently seeking participants for a clinical trial investigating Androgenetic Alopecia (AGA)—commonly known as pattern hair loss.
You do not need a formal AGA diagnosis to attend. If you suspect you have AGA, you’re welcome to screen, as long as you meet the other eligibility criteria.
Why participate?
Hair loss is common, affecting both men and women. These clinical studies aim to explore new approaches that may support future treatment options. While the main goal is to understand safety and tolerability, your participation helps shape the future of hair loss research.
What stage of hair loss are we looking for?
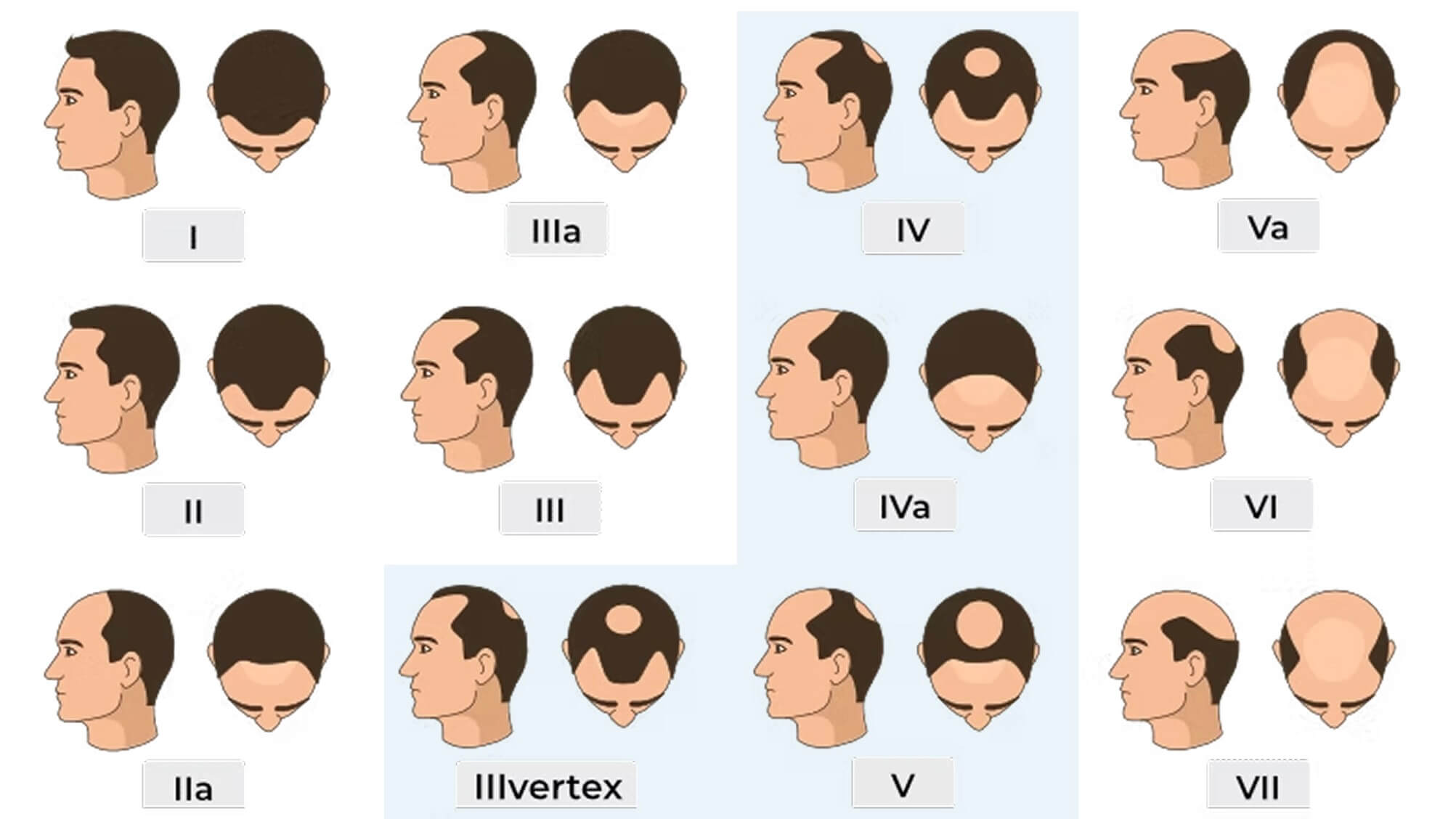
Use the guide below to see if your hair loss matches what researchers need for this study.
Find your stage
We’re specifically looking for these patterns:
Stage III Vertex
When you look down at the top of your head, you can clearly see your scalp at the crown. Your hairline has receded at the temples. There’s visible thinning where there used to be coverage.
Stage IV
Your hairline has receded significantly. The hair on your crown is sparse or minimal. There’s still a band of hair connecting the sides, but both the front and top show clear scalp visibility.
Stage IVa
Similar to Stage IV but with more pronounced frontal hair loss. The receding has progressed further at the front, while the crown shows noticeable thinning. The separating band of hair is still present but less dense.
Stage V
The hair loss has progressed—larger areas of visible scalp, the band of hair between front and back is getting narrower and less dense, and the thinning has become more pronounced overall. The areas of hair loss at the front and crown are larger and closer to connecting.
The Norwood Scale
Trial details
Additional eligibility
You may be eligible if you:
- Weigh over 60kg.
- Are experiencing male or female pattern hair loss
- Men: thinning at the crown or a receding hairline
- Have had hair loss for 12 months or more
- Are not experiencing sudden or unexplained shedding
You won’t be eligible if you:
- Have hormone-related conditions like hyperprolactinemia or prolactinoma
- Used hormone replacement therapy (HRT) in the 6 months before screening
- Have ever had:
- A hair transplant
- Microneedling, laser, or surgical scalp treatments
- Have any other scalp conditions besides AGA, including:
- Alopecia areata
- Scarring hair loss (e.g. lichen planopilaris, folliculitis decalvans)
- Lupus, psoriasis, or severe dandruff affecting the scalp
- Fungal or active scalp infections
- Any other inflammatory or scarring scalp condition
Study duration
- 4 sets of 3 overnight stays (12 nights total)
- At least 20 follow-up visits
- Up to $27,460 reimbursement for your time and commitment
Option 2:
- 4 dosing visits
- At least 18 follow-up visits
- Up to $21,860 reimbursement for your time and commitment
Are you a match?
Age
18 - 65 years old
Remuneration
Up to $27,460 *with screening reimbursement. Please see T&Cs.
Gender
Male or Female
BMI
BMI 18 - 32 kg/m2
Medical condition
Not taking any medications